Quality Procedures Manual
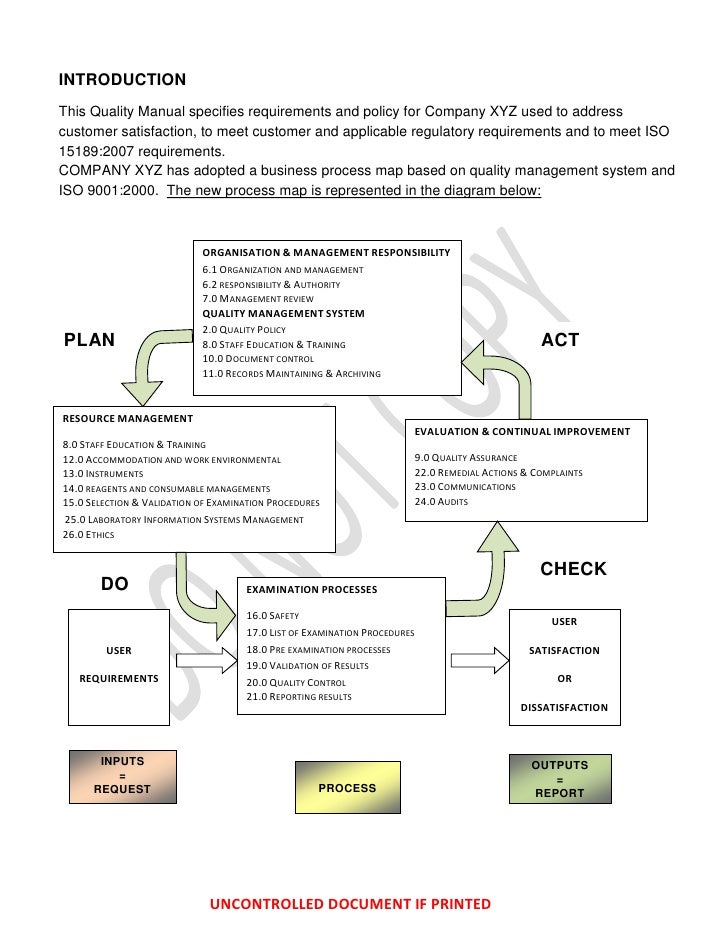
Quality manual template. Overview The quality manual template is a supplement to the laboratory quality management system training toolkit, Module 16 - Documents and records. This quality manual template is based on internationally-accepted standards, and provides guidance for public health and clinical laboratories on writing policies and procedures that support a quality management system. It comprises a main document providing information and examples to assist with writing a laboratory quality manual, and 24 appendices (examples of standard operating procedures, forms, and processes). All documents are in Word format because they are meant to serve as templates and are thus modifiable. The individual laboratories are required to customize the text of the template to the local situation. Download all the appendices.
Dec 29, 2009 - SORRY! I am not a supporter of canned quality and procedures manuals! Although the requirements of the standard are the same, the quality. Quality Procedures and Work Instructions Manual. Table of Contents. Control of Inspection, Measuring, and Test.
The six required procedures of ISO 9001 ISO 9001:2008 requires 'documented procedures' for the following six activities:. (4.2.3). Control of records (4.2.4). (8.2.2). (8.3).
Riba Quality Procedures Manual
(8.5.2). (8.5.3) Of course, these documented procedures will have to be controlled according to the first procedure listed (Control of Documents). Any more is up to you. You must determine what further documentation is required in your company for your Quality Management System to function effectively. ISO 9001 also specifies. ISO 9001:2015 ISO 9001:2015 does not require any documented procedures, and there are only three pieces of required documented information that must be ' maintained':. the scope of the quality system.

Quality Procedures Manual Wood Shops
your quality policy. your quality objectives As far as procedures go, the standard now states: 'The organization shall maintain documented information to the extent necessary to support the operation of processes' So it's up to you work out what is needed and how you want to document it. On the other hand, there is still plenty of documented information that must be ' retained' (i.e.